Пресс-центр / новости / Наука /
Комплекс-зависимая роль ацетилтрансферазы гистонов KAT8 в транскрипции и клеточном гомеостазе
Международной группой ученых при участии сотрудника ИБХ РАН Ковальчука С.И. был исследован молекулярный механизм активности фермента ацетилтрансферазы KAT8, регулирующего транскрипцию генов посредством модификации связанных с ними гистонов. Результаты работы опубликованы в журнале Molecular Cell.
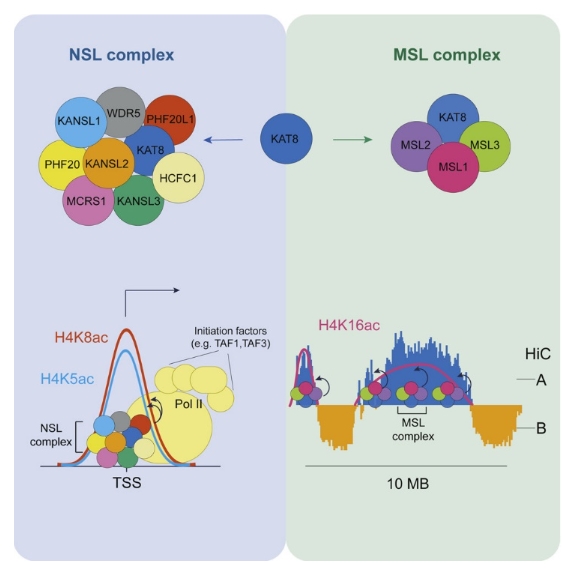
Ацетилирование Lys16 белка гистона Н4 (H4K16ac) катализируется ацетилтрансферазой гистонов KAT8. Данная модификация влияет на компактизацию ДНК – ацетилирование препятствует сворачиванию ДНК in vitro. В работе изучено влияние активности KAT8 и ацетилирования гистонаН4 на активность генов в млекопитающих, а также молекулярный механизм процесса ацетилирования при участии двух возможных комплексов NSL и MSL, содержащих KAT8. В результате исследования было показано, что специфичность KAT8 к гистонам зависит от окружения белка: ацетилирование H4K16 происходит только в составе комплекса MSL, тогда как в составе комплекса NSL белок KAT8 катализирует ацетилирование H4K5ac и H4K8ac. Кроме того, оказалось, что MSL комплекс и ацетилирование H4K16ac не являются необходимыми для пролиферации клеток и сборки хроматина, тогда как комплекс NSL является жизненно-важными для нормального функционирования клеток – его активность стимулирует инициацию транскрипции генов домашнего хозяйства. Таким образом, было продемонстрировано, что KAT8 может менять свою специфичность в зависимости от обстоятельств и проявлять сложную регуляторную активность генов в составе различных комплексов.
18 мая 2021 года


