Пресс-центр / новости / Наука /
Стрептоциннамиды А и В, депсипептиды из штамма Streptomyces sp. KMM 9044
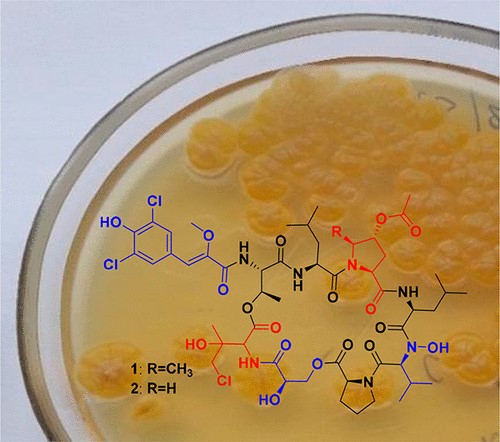
Новая структурная группа антибиотиков из нового штамма-продуцента Streptomyces sp. КММ 9044 открыта в результате совместной работы сотрудников ИБХ РАН и коллег из Тихоокеанского института биоорганической химии им. Г.Б. Елякова (ТИБОХ РАН).

Makarieva TN, Romanenko LA, Mineev KS, Shubina LK, Guglya EB, Kalinovskaya NI, Ivanchina NV, Guzii AG, Belozerova OA, Kovalchuk SI, Popov RS, Denisenko VA, Mikhailov VV, Babenko VV, Ilina EN, Malakhova MV, Terekhov SS, Kudzhaev AM, Dmitrenok PS, Yampolsky IV & Stonik VA
Штамм из коллекции морских микроорганизмов ТИБОХ РАН, который был выделен из морских отложений, собранных на северо-западном побережье Японского моря в 2009 г., был культивирован на среде с морской водой, и из культуральной жидкости выделено два соединения, обладающих антибактериальной активностью. Их структура установлена с использованием методов ядерного магнитного резонанса и хромато-масс-спектрометрии высокого разрешения, а также подтверждена серией химических трансформаций. Данное семейство соединений представляет собой хлорированные циклические депсигептапептиды, содержащие редкие и ранее не встречавшиеся в макроциклической структуре депсипептидов аминокислоты 3-гидрокси-4-хлорвалин и 4-ацетокси-5-метилпролин, а также глицериновую кислоту. Интересное свойство этих антибиотиков – сильное селективное ингибирование нескольких грам-позитивных бактерий: Micrococcus sp. (МИК 6 нг/мл, штамм KMM 1467), Arthrobacter sp. (МИК 20 нг/мл, штамм ATCC 21022) и Mycobacterium smegmatis (МИК 300 нг/мл, штамм MC2 155).
Результаты опубликованы в журнале Organic Letters.
1 июля 2022 года

