Press-room / news / Science news /
Therapeutic efficacy of antibody-drug conjugates targeting GD2-positive tumors
Both ganglioside GD2-specific immunotherapy and antibody-drug conjugates (ADCs), as a class of targeted drugs, have demonstrated clinical success as solid tumor therapies in recent years, yet practically no research has been carried out on ADCs directed to ganglioside GD2. In a new study, scientists from the Department of immunology at IBCh RAS in collaboration with colleagues from other Russian institutes for the first time show that clinically relevant anti-GD2 antibody-drug conjugates manifest potent and highly selective cytotoxicity in a wide panel of cell lines with varying GD2 expression and strongly inhibit tumor growth in mouse models of GD2-positive solid cancer.
Despite significant success in the development of antibody-drug conjugates (ADCs) for oncology indications in recent years, practically no studies explore ADCs directed at tumors expressing ganglioside GD2. In this study, ADCs were developed based on the ch14.18 antibody—the most prevalent GD2-specific therapeutic in the clinic, and the microtubule-depolymerizing agents monomethyl auristatins E (MMAE) or F (MMAF) that were conjugated to the antibody interchain cysteines by thiol-maleimide chemistry via the cleavable valine-citrulline linker. An average drug-antibody ratio (DAR) of 4 was chosen for the ADCs as a typical DAR within clinically approved conjugates. The ADCs maintained specific binding to ganglioside GD2 characteristic to the parent antibody with no cross-reactivity to other structurally similar gangliosides and showed a similar biodistribution profile to the parent antibody in the B78-D14 mouse melanoma model, reaching 8% ID/g in the tumor at 48 hours post-injection.
The GD2-specific antibody-drug conjugates showed potent and selective cytotoxic effects in a panel of tumor cell lines of human neuroblastoma, glioma, breast cancer, sarcoma, and melanoma and murine melanoma and lymphoma. The level of GD2 expression was analyzed in more than 20 cell lines of different origin, and a direct dependence of the cytotoxic effect on the level of antigen expression was observed in the cell lines for all ADCs. IC50 values reached 30 pM for the cell lines with high GD2 expression, and at the same time no cytotoxic effect was observed for GD2-negative cell lines even at high drug concentrations. Within the analyzed cell lines, ch14.18-MMAF was more effective in the cells overexpressing GD2, while ch14.18-MMAE had more prominent activity in the cells expressing GD2 at lower levels, and these differences are likely to stem from the properties of the small molecules. While MMAE and MMAF are structurally similar, MMAE is more capable of exerting the bystander killing effect – after entering target cells on antibody-mediated delivery and release in the lysosomes, it is able to penetrate into neighboring cells, leading to their death. MMAF, on the other hand, has limited ability to enter or exit the cells on its own, yet accumulates more effectively in cells with high antigen expression. Therefore, it is likely that MMAF-conjugated antibodies would be more efficient for the therapy of tumors with high GD2 expression, such as neuroblastoma, whereas MMAE-conjugated antibodies have a higher potential in tumors with lower of heterogeneous levels of GD2 expression, such as breast cancer and glioma.
Strong inhibition of tumor growth was observed for both ch14.18-MMAE and ch14.18-MMAF intravenously injected into syngeneic mouse models of GD2-positive B78-D14 melanoma and EL-4 lymphoma. For these GD2-positive models, the average tumor size in groups treated with ch14.18-MMAE or ch14.18-MMAF was several times smaller compared with the control group. In contrast to the ADCs, administration of the parent ch14.18 antibody had an insignificant effect on tumor growth inhibition. While the mouse models employed were immunocompetent, the tumors were either characterized by aggressive growth (EL-4 cells), or were immunologically “cold” (B78-D14 cells), both of these features being the case for most GD2-positive tumors. The potency of the GD2-specific ADCs in vivo indicates of their advantages over naked antibodies that are currently approved for the treatment of GD2-positive tumors.
This is the first study to analyze the efficacy of GD2-directed antibody-drug conjugates in a wide range of ganglioside GD2-expressing tumors, and its results demonstrate the prospects of the given approach in therapy.
The work is published in the Journal for ImmunoTherapy of Cancer.
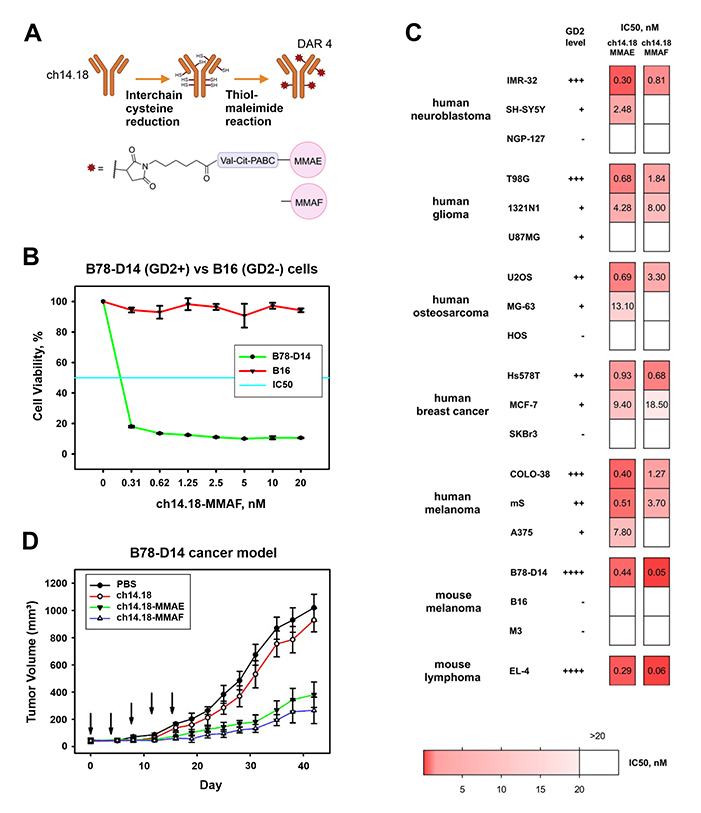
Figure 1. (A) ADC generation by thiol-maleimide chemistry. (B) Viability of GD2-positive and GD2-negative cell lines analyzed by MTT assay following incubation with ch14.18-MMAF. (C) Correlation heatmap between ganglioside GD2 expression and IC50 values in the tumor cell line panel; MTT assay following 72 h incubation with ch14.18-MMAE or ch14.18-MMAF. (D) Antitumor activity of ch14.18-MMAE, ch14.18-MMAF, and the parent antibody ch14.18 (each at 5 mg/kg body weight) in B78-D14 melanoma-bearing C57BL/6 mice.
july 19, 2022


