Пресс-центр / новости / Наука /
Различие структурно-динамических свойств и взаимодействий с липидным окружением объясняет особую функциональную роль трансмембранных доменов в подсемействе рецепторов инсулина
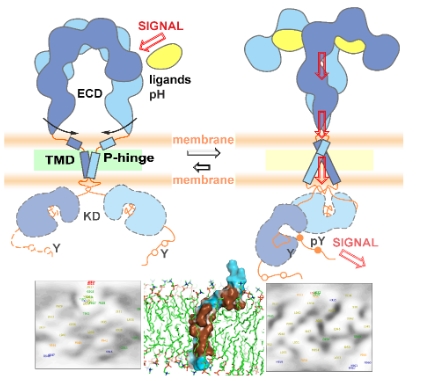
Рецепторные тирозинкиназы InsR, IGF1R и IRR подсемейства рецепторов инсулина играют важную роль в межклеточных сигнальных путях различных физиологических процессов. Имея высокую гомологию последовательности и структуры, рецепторы не схожи по своей локализации, экспрессии и функциям в организме человека. Команда ученых из ИБХ РАН в сотрудничестве с коллегами из других институтов с помощью ЯМР-спектроскопии высокого разрешения и компьютерного моделирования установила, что конформационная изменчивость трансмембранных доменов и их взаимодействие с окружающими липидами существенно различаются у представителей подсемейства.
Таким образом, в разнообразных механизмах активации InsR, IGF1R и IRR следует учитывать гетерогенное мембранное окружение. Механизм мембрано-опосредованного контроля передачи сигналов рецепторами открывает новые перспективы для разработки таргетных методов лечения заболеваний, связанных с дисфункцией данных рецепторов.
Результаты опубликованы в журнале International Journal of Molecular Sciences.
28 февраля 2023 года


