Пресс-центр / новости / Наука /
Внутримолекулярная водородная связь в N6-замещенных 2-хлораденозинах по данным ЯМР-спектроскопии
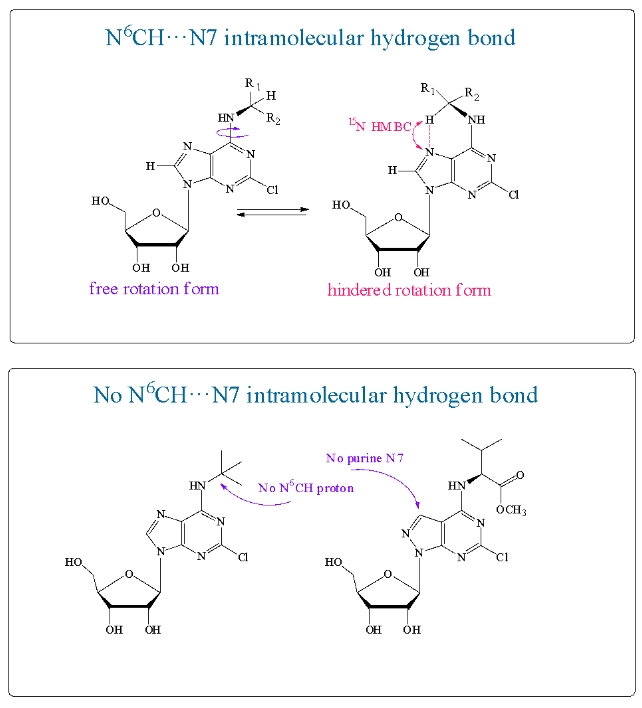
В спектрах ЯМР N6-замещенных 2-хлораденозинов были обнаружены две формы. Доля мини-формы составляла 11–32% от основной. Мини-форма характеризуется отдельным набором сигналов в спектрах ЯМР. Команда ученых из ИБХ РАН предположила, что она возникает за счет внутримолекулярной водородной связи между протоном N6-CH заместителя и атомом N7 пурина. Спектр 15N-HMBC подтвердил наличие водородной связи в мини-форме нуклеозида и ее отсутствие в основной форме. Были синтезированы соединения, в которых образование такой водородной связи невозможно. В них отсутствовал либо атом N7 пурина, либо протон N6–CH заместителя. В спектрах ЯМР этих нуклеозидов мини- форма не обнаружена, что подтверждает необходимость образования внутримолекулярной водородной связи для появления мини-формы.
Работа опубликована в журнале International Journal of Molecular Sciences.
14 июня 2023 года


