Пресс-центр / новости / Наука /
Галектин-9 как потенциальный модулятор адгезии лимфоцитов к эндотелию посредством связывания с гликаном Н системы групп крови АВН
Адгезия лейкоцитов — ключевой этап их проникновения в воспалительный очаг. Этот процесс осуществляется благодаря взаимодействию интегринов, селектинов и CD44, в то время как роль галектинов не до конца ясна. Известно, что галектины способны взаимодействовать с олиголактозаминами эндотелиальных клеток, а также, что in vitro галектины тандемного типа (гал-4, -8 и -9) связываются с с высокой аффинностью с гликанами системы крови АВН. В этой работе на клеточных линиях показано, что гал-9 опосредует адгезию лимфоцитов к эндотелиальным клеткам, благодаря связыванию с их Н-гликаном, и это позволяет предположить, что адгезия лимфоцитов к эндотелию в кровотоке происходит аналогично и регулируется степенью экспрессии галектина-9.
Наряду с интегринами, селектинами и CD44 лейкоцитов, потенциальными участниками их адгезии к эндотелию могли бы быть и белки семейства галектинов (гал), широко представленные на лимфоцитах. Галектины связываются с олиголактозаминами (LN), которые есть на эндотелиальных клетках (ЭК). Помимо LN, на ЭК мощно экспрессируются гликаны системы группы крови АВH, которые находятся в составе гликолипидов и гликопротеинов. Однако, информация о том, что галектины вовлечены в адгезию к эндотелию благодаря связыванию именно с АВН-гликанами, отсутствует, хотя галектины тандемного типа (гал-4, -8 и -9) узнают их с аффинностью на порядок выше, чем LNs. Гал-8 и гал-9 выявляются в гликокаликсе, они имеют два углеводсвязывающих домена (УСД) - N-УСД и C-УСД. Первый проявляет высокое сродство к LN и вовлечен в заякоривание лектина на клетке, с помощью второго галектины связываются с гликанами других клеток, в частности с АВН-гликанами. В этой работе исследовано, способен ли гал-9 опосредовать адгезию лимфоцитов к эндотелиальным клеткам, благодаря связыванию с их Н-гликаном. Модельной системой служило взаимодействие клеток Jurkat (лимфоцитарного происхождения, экспрессируют гал-9) с эндотелиальной клеточной культурой EA.hy 926 (экспрессирует Н-гликаны). В фокусе исследования был Н-антиген, так как в клетках EA.hy 926 отсутствуют A- и B-гликозилтрансферазы. Клетки EA.hy 926 дефукозилировали, затем в них встраивали синтетический липидный конструкт, содержащий в качестве гликана трисахарид Fucα1-2Galβ1-4GlcNAcβ (Н (тип 2) (рис. 1).
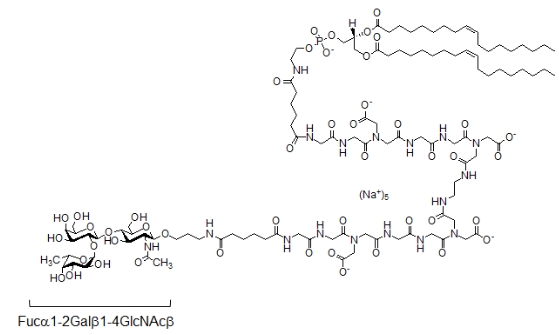
Рисунок 1. Структура синтетического гликолипида H (тип 2)
После встраивания, гликан выявлялся в непосредственной близости от DPH-окрашенных мембранных зон и совпадал по расположению с Н-гликаном в нативных клетках (рис. 2).
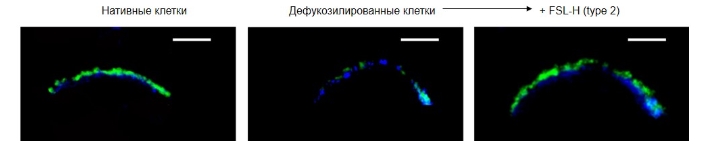
Рисунок 2. Локализация H (тип 2) в клетках EA.hy 926 cells, конфокальная микроскопия. Для окрашивания мембраны использовали реагент DPH (1,6-дифенилгексатриен, синий цвет)), который накапливается и флуоресцирует только в гидрофобной среде. Н-гликан выявляли с помощью биотинилированного лектина UEA I и конъюгата стрептавидина с FITC (зеленый цвет). Масштаб=5 мкм.
Удаление фукозы приводило к уменьшению связывания свободного гал-9 с клетками. Благодаря встраиванию H (type 2), связывание гал-9 с дефукозилированными клетками увеличивалось в три раза (рис. 3), и было сопоставимо с таковым для нативных клеток.
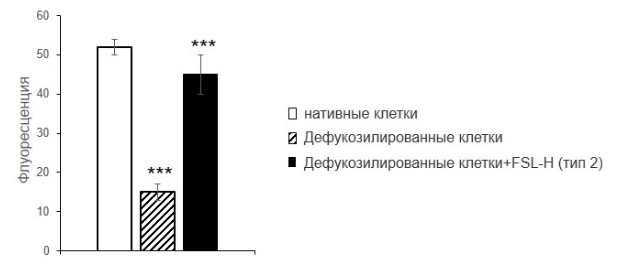
Рисунок 3. Связывание гал-9 с клетками EA.hy 926, цитофлуориметрия. Клетки инкубировали с фукозидазой, затем – с гликолипидом H (type 2). После встраивания и отмывки клетки инкубировали с гал-9, антителами к гал-9 и вторичным конъюгатом IgG-FITC. Результаты цитофлуориметрического анализа представлены как среднее трех независимых экспериментов, p<0,001.
Наличие трисахарида H (type 2) оказалось достаточным для взаимодействия гал-9 в составе клеток Jurkat cell с клетками EA.hy 926. Для исследования межклеточной адгезии клетки Jurkat добавляли к монослою ЭК, содержащих разное количество Н-гликана: к нативным, дефукозилированным и дефукозилированным с последующим встраиванием в них H (тип 2). После отмывки, прилипшие к EA.hy 926 Jurkat выявляли, идентифицируя на них гал-9; клетки, экспрессирующие лектин подсчитывали. На обработанных фукозидазой EA.hy 926 клетках выявлялось всего 4% от количества Jurkat, прилипших к нативным клеткам (рис. 4 Б, Г), а на дефукозилированных клетках после встраивания H (type 2) - до 50% (рис. 4, В, Г).
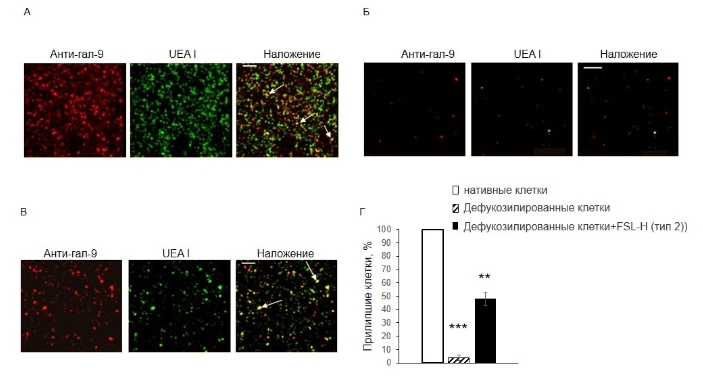
Рисунок 4. Адгезия клеток Jurkat к интактным (А), дефукозилированным (Б) и дефукозилированным клеткам EA.hy 926 после встраивания H (type 2) (В), конфокальная микроскопия. Гал-9 детектировали анти-гал-9+ IgG-Alexa594 (красный), H (type 2) детектировали биотинилированным UEA I + Str-FITC (зеленый). Клетки Jurkat, прилипшие к EA.hy 926, на картинках, полученных наложением красного и зеленого флуоресцентного сигнала (желтый), показаны стрелками. Прилипшие клетки, подсчитанные в трех независимых измерениях, представлены как процент адгезированных клеток к интактным (Г). *** p<0.001, ** p<0.01. Масштаб: 200 мкм.
Таким образом, в этой работе показано, что гал-9 опосредует адгезию клеток Jurkat к EA.hy 926, связываясь с Н-гликаном последних. С учетом того, что уровень экспрессии галектинов в гликокаликсе лимфоцитов тонко регулируется, найденный в модельной системе механизм, по-видимому, задействован и в адгезии лимфоцитов крови к эндотелию.
Работа опубликована в журнале Biomolecules.
25 сентября 2023 года


