Пресс-центр / новости / Наука /
Возрастные изменения в коре головного мозга человека приводят к изменению функций митохондрий и атрофии астроцитов
Команда ученых из ИБХ РАН, совместно с коллегами из других институтов, изучила сложные клеточные изменения, происходящие с возрастом в головном мозге человека. Исследование проводилось на ткани коры головного мозга человека в возрасте от 22 до 72 лет и дало уникальную информацию о влиянии старения на астроциты и нейроны человека.
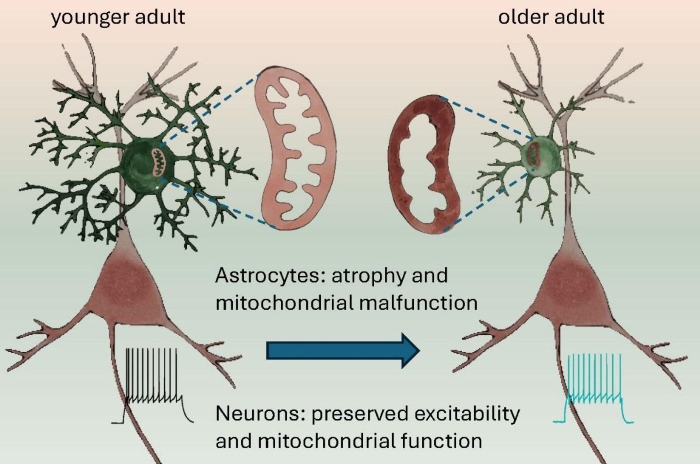
Исследование, опубликованное в журнале Nature Communications, бросает вызов существующим парадигмам, показывая, что старение по-разному влияет на астроциты и нейроны. С возрастом происходит значительное снижение количества восстановленных цитохромов в астроцитах, что говорит об изменениях функций митохондрий. Примечательно, что в нейронах такого явления не наблюдалось, что подчеркивает клеточную специфичность процесса старения. Исследование выявляет изменения в соотношении белков и липидов в астроцитах и нейронах, подчеркивая сложность клеточных изменений, связанных со старением.
С возрастом также происходит атрофия астроцитов уменьшается количество их веточек, снижается объемная доля листочков, сокращается астроцитарный домен. Эти изменения коррелируют с функциональными изменениями, включающими в себя потерю связи между астроцитами через щелевые контакты и повышение входного сопротивления данных клеток.
Морфологические изменения коррелируют с увеличением количества глиального фибриллярного кислого белка (GFAP), и уменьшением эзрина, белка расположенного в астроцитарных листочках. Удивительно, но эти изменения в астроцитах не сопровождались значительными изменениями возбудимости нейронов или спонтанной тормозной постсинаптической передачи сигналов.
Представленное исследование уникально еще и потому, что результаты получены на клетках мозга человека, а не на животных. Полученные данные открывают путь к более глубокому пониманию процесса старения, учитывая реакции, специфичные для каждого типа клеток в активной среде мозга.
25 декабря 2023 года


