Пресс-центр / новости / Наука /
Определены конформационные изменения рецепторной тирозинкиназы IRR при активации
Сотрудники Лаборатории клеточной биологии рецепторов ИБХ РАН совместно с коллегами из ИФХЭ им. А.Н. Фрумкина РАН и ИК им. А.В. Шубникова РАН провели исследование структуры IRR методами атомно-силовой микроскопии и малоуглового рентгеновского рассеяния. Определены конформации рецептора в активном и неактивном состоянии, на основании полученных данных предложен механизм активации.

, , Mozhaev AA, Goryashchenko AS, , Orsa AN, Bocharov EV, Deyev IE, Petrenko AG

, , Mozhaev AA, Deyev IE, , , Goryashchenko AS, Bocharov EV, , , , Petrenko AG
Для активации большинства рецепторных тирозинкиназ необходим лиганд белково-пептидной природы, способный взаимодействовать с внеклеточной частью рецептора. Взаимодействие происходит одновременно в нескольких местах, приводя к димеризации и активации. Особенным является семейство рецептора инсулина, члены которого существуют изначально в виде димеров, связанных дисульфидными связями. При взаимодействии их внеклеточной части с лигандом, они меняют свою конформацию и активируются. В это семейство входит три рецептора: рецептор инсулина (Insulin receptor, IR), рецептор инсулиноподобного фактора роста (Insulin-like growth factor 1 receptor, IGF-1R) и рецептор, подобный рецептору инсулина (Insulin receptor-related receptor, IRR). Рецепторы данного семейства имеют одинаковую доменную организацию, высокогомологичны между собой и, предположительно, имеют близкие механизмы функционирования. Активация рецептора инсулина модулирует жизненно важные метаболические процессы, а активация рецептора инсулиноподобного фактора роста регулирует процессы клеточного роста и деления.
Авторами ранее показано, что IRR представляет собой сенсор внеклеточной щелочной среды и принимает участие в регулировании кислотно-щелочного баланса в организме. Определение молекулярного механизма активации IRR представляет особый интерес для понимания фундаментальных основ механизма щелочной чувствительности, а также для развития в будущем новых терапевтических подходов и созданию лекарств для лечения заболеваний, связанных с нарушением кислотно-щелочного равновесия, патологий клеточного деления и дифференцировки, в частности, некоторых форм рака.
Методами атомно-силовой микроскопии высокого разрешения и малоуглового рентгеновского рассеяния провели исследование структуры IRR. Полученные данные показывают, что в среде с нейтральным значением pH структура неактивного IRR имеет две конформации, одна из которых симметрична и схожа с неактивной Λ / U - формой эктодоменов IR и IGF-1R, тогда как вторая каплевидная и асимметричная. В среде со щелочным значением pH обнаружены Γ - и T – образные конформации активного рецептора, обе продемонстрировали образование комплекса внутриклеточных каталитических доменов, ответственных за аутофосфорилирование. Существование двух активных форм коррелирует с ранее описанной положительной кооперативностью активации IRR.
Опубликованные результаты исследования являются впервые полученными данными о пространственной структуре IRR, на основании полученных данных предложена модель механизма активации рецептора. Данные также могут иметь ценность для интерпретации результатов структурных исследований IR и IGF-1R.
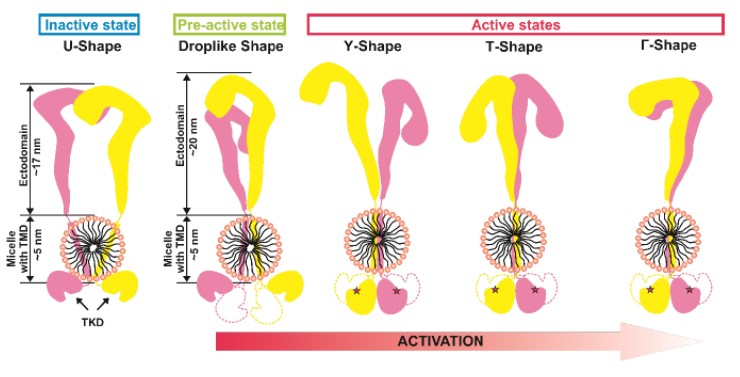
Рисунок 1. Диаграмма, показывающая наблюдаемые формы IRR и модель его активации.
TMD - трансмембранный домен, TKD - тирозинкиназный домен.
Работа выполнена при поддержке проектов Российского фонда фундаментальных исследований 20-04-00959, 17-00-00486, Министерства Науки и Высшего Образования РФ и опубликована в двух статьях Journal of Biological Chemistry [1] [2].
5 апреля 2021 года

