Пресс-центр / новости / Наука /
Гены скейлеры - регуляторы масштабной инвариантности эмбрионального развития
Эмбрионы многих организмов способны сохранять инвариантность своей структуры независимо от размера - т.н. явление эмбрионального скейлинга. Например, эмбрионы морского ежа или лягушки, развившиеся из отдельных клеток, изолированных вскоре после начала дробления яйца, регулируют свою структуру так, что выглядят как уменьшенные копии нормальных. Сотрудники Лаборатории молекулярных основ эмбриогенеза ИБХ РАН впервые предложили общий подход к изучению механизмов эмбрионального скейлинга. Они предсказали и математически доказали существование специальных генов-скейлеров, экспрессия которых зависит от размеров эмбрионов, разработали метод их прицельного поиска и описали на примере одного из найденных скейлеров шпорцевой лягушки, гена mmp3, неизвестный ранее механизм размер-зависимой регуляции эмбрионального паттернинга.
Проблема удивительной способности эмбрионов многих животных регулировать свою структуру пропорционально размеру, т.н. явление эмбрионального скейлинга, является одной из самых важных, но малоизученных в биологии. В научной литературе этот феномен впервые был описан немецким эмбриологом и философом Гансом Дришем более 120 лет назад. В опытах на эмбрионах морского ежа Дриш неожиданно обнаружил, что из отдельных клеток, изолированных сразу же после первого или второго деления дробления оплодотворенного яйца или даже из половинок эмбриона на стадии бластулы, разрезанного пополам, во многих случаях развивались совершенно правильные, но уменьшенные копии нормальных эмбрионов (Рис. 1A). В дальнейшем аналогичные примеры скейлинга были описаны разными авторами на эмбрионах многих других животных (Рис. 1B и C). В настоящее время стало понятно, что данный феномен, завораживающий исследователей на протяжении века, представляет собой наиболее яркое проявление фундаментальной особенности живых организмов - использовать на систематической основе неравновесную самоорганизацию для своего крупномасштабного структурирования. Несмотря на то, что в некоторых случаях конкретных молекулярные механизмы эмбрионального скейлинга были установлены, общего решения для данной фундаментальной проблемы, лежащей на стыке молекулярной биологии, физики и математики, не существовало.
В только что опубликованной работе сотрудники Лаборатории молекулярных основ эмбриогенеза ИБХ РАН предложили такой подход, постулировав существование специальных генов-скейлеров, уровень экспрессии которых зависит от размеров эмбрионов (Рис. 1D). Авторы предположили, что концентрации белков, кодируемых этими генами, играют роль датчиков, регулирующих в зависимости от размера эмбриона масштаб морфогенетических градиентов. Как известно, такие диффузионные градиенты сигнальных белков-морфогенов возникают по механизму неравновесной самоорганизации и служат для пространственной разметки крупномасштабной структуры эмбриона.
Проанализировав in silico около полумиллиона вариантов различных математических реакционно-диффузионных моделей самоорганизации морфогенетических градиентов, авторы выяснили, что в том случае, когда такая модель способна воспроизводить скейлинг градиента при изменении размера реактора, она действительно всегда имеет в своем составе белок, концентрация которого при этом резко меняется. Такие белки, играющие роль датчиков размера и названные авторами скейлерами, необходимы для правильного скейлинга морфогенетических градиентов. На основе полученных данных, был разработан метод прицельного поиска генов, кодирующих скейлеры, у реальных эмбрионов. В результате на экспериментальной модели эмбрионов шпорцевой лягушки авторы выявили гены, которые действительно резко меняли свою экспрессию в ответ на изменение размера эмбриона (Рис. 1D). Интересно, что число таких генов оказалось крайне небольшим (около двадцати). Для одного из них, гена секретируемой металлопротеиназы 3 (Mmp3), впервые был описан неизвестный ранее молекулярный механизм, позволяющий подстраивать под размер эмбриона масштаб диффузионных градиентов морфогенных белков Chordin и Noggin1/2 (Рис. 1D). В период раннего развития именно эти градиенты определяют разметку клеточных территорий, дифференцирующихся затем в основные зачатки эмбриональной оси тела - нервную систему, хорду, сомитную мезодерму и вентральную мезодерму (Рис. 1D).
Разработанный подход может быть использован и в любых других случаях, когда необходимо изучить заранее неизвестные механизмы эмбрионального скейлинга у самых разных модельных организмов. Работа поддержана грантом РНФ №19-14-00098 и опубликована в журнале Developmental Cell.
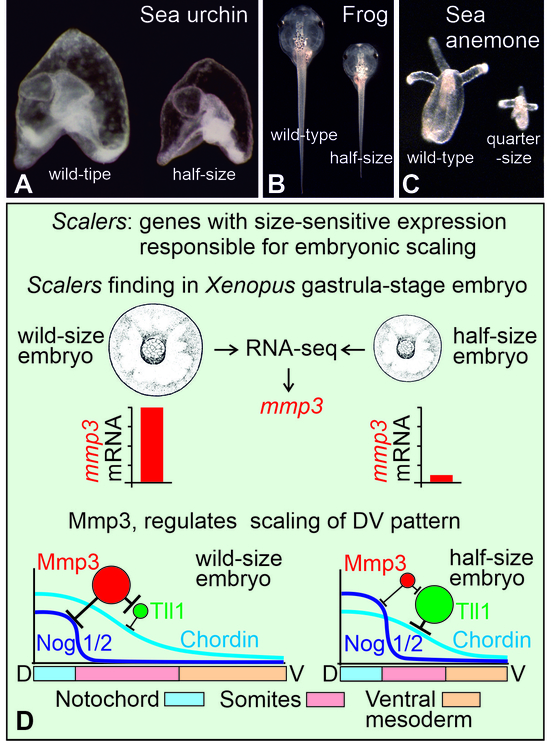
Рис. 1. Эмбриональный скейлинг и регулирующие его гены-скейлеры.
A и B. Личинки морского ежа Strongylocentrotus droebachiensis и головастики шпорцевой лягушки Xenopus laevis развившиеся из эмбрионов дикого типа (слева на каждом фото) и из единичных клеток, выделенных из 2х-клеточных эмбрионов (справа на каждом фото). С. Полипы актинии Nematostella vectensis, развившиеся из эмбриона дикого типа (слева) и из единично клетки, выделенной из 4х-клеточного эмбриона (справа) (фото Фёдора Ерошкина). D. Графическая схема, суммирующая результаты статьи.
10 января 2022 года


