Пресс-центр / новости / Наука /
Генетически кодируемая система, основанная на биолюминесцентном резонансном переносе энергии, для фотодинамической терапии глубинных опухолей
Разработана генетически кодируемая BRET-пара «внутренний источник света-фотосенсибилизатор» NanoLuc-miniSOG, которая, будучи нацеленной на раковые клетки, позволяет проводить BRET-активированную ФДТ для лечения глубинных опухолей.
Фотодинамическая терапия (ФДТ) является малоинвазивным методом лечения рака, основанным на активированной светом противоопухолевой активности фотосенсибилизаторов. Однако ограниченная глубина проникновения света в ткани не позволяет применять этот метод для лечения глубинных опухолей, и, таким образом, затрудняет широкое клиническое внедрение ФДТ. Сотрудники Отдела иммунологии и Отдела пептидно-белковых технологий совместно ИБХ РАН с коллегами из НПП «Питомник лабораторных животных», ИОФ РАН и НИЯУ МИФИ разработали генетически кодируемую систему, основанную на биолюминесцентном резонансном переносе энергии (BRET) для применения в ФДТ. Данная система содержит так называемый «внутренний» источник света (люциферазу NanoLuc) и фотосенсибилизатор (фототоксичный белок miniSOG). NanoLuc-miniSOG представляют собой BRET-пару, в которой окисленная форма люциферазного субстрата является донором энергии для возбуждения miniSOG. Находясь в возбужденном состоянии, флавопротеин miniSOG генерирует АФК. Будучи генетически кодируемой, данная конструкция может быть доставлена к опухолям, расположенным практически на неограниченной глубине. На мышах с привитыми опухолями, экспрессирующими ген NanoLuc-miniSOG, мы показали возможность элиминации опухоли при введении животным люциферазного субстрата. Используя псевдотипированные лентивирусы, специфичные к HER2-онкомаркеру, мы продемонстрировали возможность адресной доставки созданной генетической конструкции непосредственно в опухолевые клетки in vivo, при этом коэффициент ингибирования роста опухоли (TGI) составил более 67%. Таким образом, проведенное исследование является доказательством возможности проведения ФДТ глубинных новообразований без внешнего источника света.
Работа опубликована в журнале Light: Science & Applications.
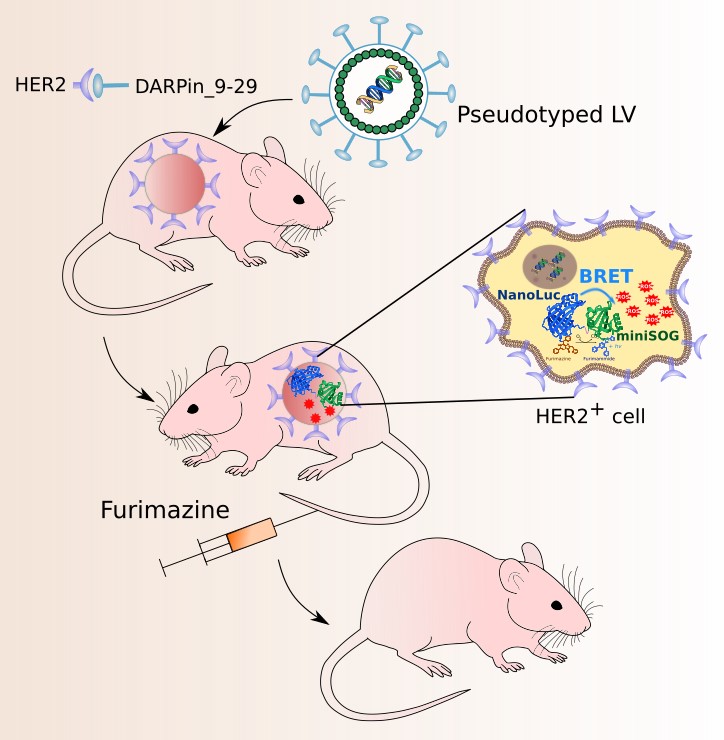
Направленная ФДТ с использованием HER2-специфичных лентивирусов (LV) в качестве носителей гена NanoLuc-miniSOG. HER2-специфичные LV, несущие ген NanoLuc-miniSOG, взаимодействуют с рецептором HER2 на поверхности раковых клеток. Узнавание онкомаркера LV-частицами возможно благодаря встроенному в оболочку лентивируса HER2-специфичного скаффолда DARPin_9-29. Гены люциферазы NanoLuc и фототоксичного белка miniSOG представляют единую генетическую конструкцию. Энергия, генерируемая субстратом люциферазы фуримазином при его окислении NanoLuc, безызлучательно передается miniSOG, что приводит к возбуждению фотосенсибилизатора и образованию АФК, которые, в свою очередь, убивают раковые клетки.
16 марта 2022 года


