Пресс-центр / новости / Наука /
Синтез новых 5'-норкарбоциклических аза/дезазапуриновых флексимеров - неконкурентных ингибиторов пуриннуклеозидфосфорилазы E. coli
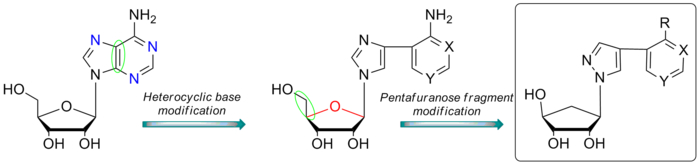
Сотрудниками Лаборатории биосинтеза низкомолекулярных физиологически активных соединений и Лаборатории биофармацевтических технологий (ИБХ РАН) совместно с коллегами из Института молекулярной биологии им. В. А. Энгельгардта синтезирована новая серия флексимерных 5'-норкарбоциклических аза/дезаза-пуриновых аналогов нуклеозидов и исследована их ингибирующая способность по отношению к пуриннуклеозидфосфорилазе E. coli.
Серия 5'-норкарбоциклических аза/деаза-пуриновых аналогов нуклеозидов была синтезирована из 6-оксибицикло[3.1.0.]гекс-2-ена и пиразолсодержащих флексимерных аналогов гетероциклических оснований по методике Троста. Три соединения оказались неконкурентными ингибиторами с константами ингибирования 14–24 мМ. На основании полученных данных можно предположить, что новые 5'-норкарбоциклические аналоги нуклеозидов взаимодействуют с активным центром пуриннуклеозидфосфорилазы подобно природным гетероциклическим основаниям. При этом наличие циклопентильного фрагмента с 2'- и 3'-гидроксильными группами необходимо для ингибирующих свойств, так как соединения без этих групп не проявляли ингибирующего действия в условиях эксперимента.
Результаты исследования опубликованы в журнале Frontiers in Chemistry (IF 5.545).
11 июля 2022 года


