Пресс-центр / новости / Наука /
Новые структурные данные позволили изучить механизм ингибирования онкоканала TRPV6 природным фитоэстрогеном генистеин
В организме человека Ca2+ - селективный ионный канал TRPV6 является ключевым участником клеточной пролиферации, его избыточная экспрессия наблюдается при самых тяжелых видах рака, включая лейкемию, рак молочной железы, предстательной железы, толстой кишки, яичников, щитовидной железы и эндометрия. В то время как значительные усилия исследователей были направлены на разработку синтетических ингибиторов TRPV6, блокаторы природного происхождения, несмотря на свою фармакологическую ценность, до сих пор оставалась вне поля зрения исследователей.
В своей совместной работе сотрудники Лаборатории моделирования биомолекулярных систем ИБХ РАН и исследователи из Колумбийского университета (Нью-Йорк, США) выявили структурные аспекты взаимодействия человеческого TRPV6 с природным фитоэстрогеном генистеин, распространённым в различных видах бобовых растений (в т.ч. в соевых бобах). Ранее в клинических исследованиях было показано, что диетический генистеин обладает целым рядом потенциально полезных эффектов, включая способность ингибировать клеточную инвазию и метастазирование при различных формах рака человека.
В настоящей работе с помощью совместного применения новейших методов криоэлектронной микроскопии, электрофизиологии, кальциевого имиджинга, белковой инженерии и молекулярного моделирования коллективу ученых удалось показать, что генистеин, связываясь с TRPV6, работает как поровый блокатор, модифицирующий структуру и свойства активационных ворот ионного канала. Генистеин связывается с порой TRPV6 в двух местах (см. Рис. 1): в сайте 1 - благодаря образованию водородных связей с остатками L571 диагонально расположенных субъединиц, и в главной позиции сайта 2, взаимодействия с несколькими полярными группами белка. При этом происходит изменение конформации образующих пору трансмембранных α-спиралей, нарушающее симметрию структуры поры и модифицирующее активационные ворота канала. В результате канал переходит в непроводящее состояние. Генистеин в главной позиции сайта 2 стабилизируется еще одной молекулой генистеина во вторичной позиции, где она локализуется с помощью π-стэкинг взаимодействия с остатками W583. Весь комплекс закрытой поры TRPV6 дополнительно стабилизируется ионами Zn2+ .
Молекулярный механизм поровой блокады TRPV6 генистеином уникален среди всех ранее описанных механизмов ингибирования TRP-каналов природными и синтетическими антагонистами. Учитывая высокий потенциал химического синтеза генистеина и его производных, полученные результаты открывают новые возможности для рационального конструирования лекарственных соединений, нацеленных на TRPV6, включая новые противораковые препараты.
Работа опубликована в журнале Nature Communications.
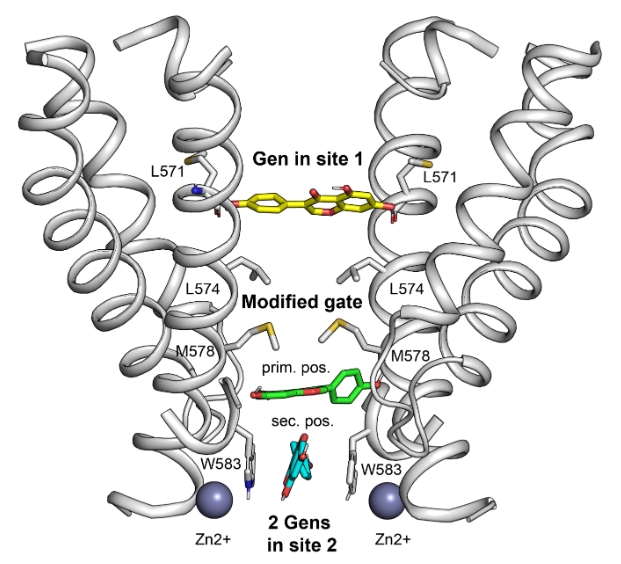
Рис.1. Пора TRPV6, блокированная генистеином (Gen). Трансмембранные α-спирали белка, образующие пору, показаны в ленточном представлении (изображены две субъединицы из четырёх). Молекула генистеина в сайте 1 показана желтым цветом; в сайте 2, в главной позиции – зеленым, во вторичной позиции – голубым. Сферами изображены ионы Zn 2+ . Между сайтами 1 и 2 расположены активационные ворота канала, свойства которых меняются при связывании генистеина.
23 мая 2023 года


