Пресс-центр / новости / Наука /
Эндогенный ретровирус регулирует ген, важный для работы мозга
Фенотип определяется не только набором генов, но и регуляторными областями генома, формирующими профиль генетической экспрессии в различных тканях. В исследовании, проведенном в двух лабораториях ИБХ, а также нескольких других учреждениях, показано влияние провируса, встроенного в ДНК человека, на экспрессию важного для работы мозга гена, отвечающего за синтез нейромедиаторов. Не исключено, что это сыграло существенную роль в эволюции человека.
Изучение генетических особенностей, отличающих людей и их ближайших родственников — шимпанзе, — не только интересно с эволюционной точки зрения, но и может пролить свет на этиологию некоторых заболеваний человека. Учитывая практически полную идентичность кодирующих белок генов у высших приматов, принято считать, что основные отличия обусловлены различной регуляцией работы генов (например, в результате мутаций в регуляторных областях ДНК), нежели генами как таковыми. С этой точки зрения мобильные элементы как источники геномной нестабильности, обладающие значительным регуляторным потенциалом, вызывают большой интерес.
Ученые из группы геномного анализа сигнальных систем клетки и Лаборатории молекулярных основ эмбриогенеза Института биоорганической химии РАН и нескольких других научных учреждений Москвы заинтересовалась поиском и изучением эндогенных ретровирусов (ЭРВ) — «самокопирующихся» последовательностей ДНК, которыми изобилует наш геном. Особенное внимание привлекли ЭРВ семейства HERV-K (HML-2) — одна из четырех групп ретроэлементов, которые были активны на момент расхождения предковых линий человека. Работа опубликована в журнале Proceedings of National Academy of Sciences U.S.A. [1] осенью 2013 года.
Среди представителей группы ретроэлементов HERV-K (HML-2) обнаружено 133 копии, специфичные только для генома человека. Выбор пал на ЭРВ еще и потому, что расположение регуляторных элементов, необходимых для транскрипции вирусной РНК (таких как промотор, энхансер и сигнал полиаденилирования), точно известно, и все они сосредоточены в определенных участках провируса — а именно, длинных концевых повторах (long terminal repeat, LTR). И эти регуляторные элементы могут служить не только нуждам ретровируса, но и оказывать влияние на гены, находящиеся поблизости.
Для изучения энхансерной активности длинных концевых повторов было отобрано шесть вставок ЭРВ, находящихся на расстоянии до пяти тысяч пар нуклеотидов от промоторов известных генов человека (считалось, что на таком расстоянии энхансерный эффект может проявиться с наибольшей вероятностью). Способность LTR усиливать активность промоторов соседних генов изучали в экспериментах с использованием репортерных конструкций. Обнаружилось, что только три из шести исследуемых LTR существенно увеличивали экспрессию репортерного гена. И лишь в одном случае из трех наблюдалась корреляция между энхансерной активностью LTR в экспериментах с репортерным геном и экспрессией соседнего гена. Им оказался ген PRODH — подробнее о нем будет сказано ниже.
Кроме того, в клетках, в которых наблюдалась наибольшая энхансерная активность, эндогенная копия LTR была гипометилирована, то есть находилась в активном состоянии. Это означает, что наблюдаемый в репортерных экспериментах энхансерный эффект может проявляться и в естественных условиях и быть причиной высокой транскрипционной активности соседнего гена. Как можно догадаться, в случае с шимпанзе вставка эндогенного ретровируса перед геном PRODH отсутствует, и эксперименты с репортерными конструкциями подтверждают, что промотор шимпанзе значительно уступает по силе человеческому, содержащему ретровирусный энхансер.
Ген PRODH, оказавшийся под влиянием ретровирусного энхансера, кодирует ключевой фермент катаболизма пролина — пролиндегидрогеназу. Согласно литературным данным, этот ген необходим для нормального функционирования центральной нервной системы: мутации или делеции этого гена связывают с риском развития шизофрении и других психических нарушений. Это можно объяснить тем, что в нервной системе фермент PRODH может участвовать в синтезе нейромедиаторов — например, глутамата. Исследования транскрипционной активности показали, что наиболее активно PRODH транскрибируется в мозге, и особенно в гиппокампе. В этом же отделе LTR-энхансер был гипометилирован. А в экспериментах с первичными культурами нейрональной ткани было показано, что промотор и ретровирусный энхансер гена PRODH обеспечивают нейрон-специфическую экспрессию репортерного флуоресцентного гена, — то есть LTR является тканеспецифическим энхансером и способен активировать экспрессию гена PRODH в нервной ткани. Стоит также отметить, что транскрипционная активность PRODH в мозге человека в среднем выше, чем у шимпанзе, что скорее всего является результатом появления такого энхансера.
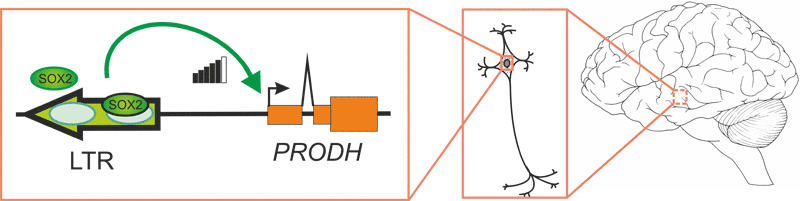
Схема транскрипционной регуляции гена PRODH ретровирусным энхансером. Человек-специфичный энхансер, образованный в результате встраивания эндогенного ретровируса, активирует промотор гена PRODH в мозге. Энхансерная активность длинного концевого повтора ЭРВ обусловлена связыванием с транскрипционным фактором SOX2.
Кроме всего прочего обнаружилось, что LTR функционирует как энхансер вместе с внутренним регуляторным элементом гена PRODH — CpG-островком (см. рисунок). При этом энхансерная активность LTR подавляется метилированием, что подтверждается экспериментами in vitro и данными о характере метилирования LTR в различных тканях. Другими словами, там, где нет необходимости в повышенной транскрипции PRODH, этот энхансер бывает «выключен» с помощью метилирования, и наоборот. В отличие от LTR, CpG-островок был гипометилирован во всех исследованных образцах, независимо от уровня экспрессии PRODH. Следовательно, «включение» CpG-островка — важное, но не достаточное условие для обеспечения высокой транскрипционной активности гена.
Выступление первого автора публикации в PNAS — Марии Сунцовой — на семинаре Совета молодых ученых ИБХ, прошедшем 19 декабря 2013 года.
Чтобы понять, какие транскрипционные факторы (ТФ) участвуют в регуляции энхансерной активности LTR, в его нуклеотидной последовательности был проведен поиск мотивов узнавания ТФ; там обнаружилось два сайта связывания с фактором SOX2 (см. рисунок). И действительно: добавление SOX2 ведет к усилению энхансерного эффекта. Стоит отметить, что SOX2 экспрессируется на высоком уроне именно в гиппокампе, где он может связываться с LTR.
Таким образом, встраивание энхансерно-активного эндогенного ретровируса перед геном PRODH могло активировать его экспрессию, что, в свою очередь, могло повлиять на баланс нейромедиаторов в мозгу. И это могло отразиться на развитии центральной нервной системы и поведении в ходе эволюции человека.
По мотивам публикации в PNAS на сайте «Элементы» размещена научно-популярная статья [2].
Литература
-
Suntsova M., Gogvadze E.V., Salozhin S., Gaifullin N., Eroshkin F., Dmitriev S.E., Martynova N., Kulikov K., Malakhova G., Tukhbatova G., Bolshakov A.P., Ghilarov D., Garazha A., Aliper A., Cantor C.R., Solokhin Y., Roumiantsev S., Balaban P., Zhavoronkov A., Buzdin A. (2013). Human-specific endogenous retroviral insert serves as an enhancer for the schizophrenia-linked gene PRODH. Proc. Natl. Acad. Sci. U.S.A. 110,
19472–19477; - Элементы: «Вирус, встроившийся в геном наших предков, влияет на работу мозга».
17 декабря 2013 года

