Пресс-центр / новости / Наука /
Создан новый подход к персонифицированной иммунотерапии Т-клеточных лимфом
Введение Т-клеток, модифицированных химерными антигенными рецепторами (CART), является одним из наиболее активно развивающихся направлений иммуноонкологии. За последнее десятилетие данная область клеточной терапии совершила значительный скачок от оптимизации структуры химерных антигенных рецепторов и экспериментов на модельных животных до успешного клинического применения.
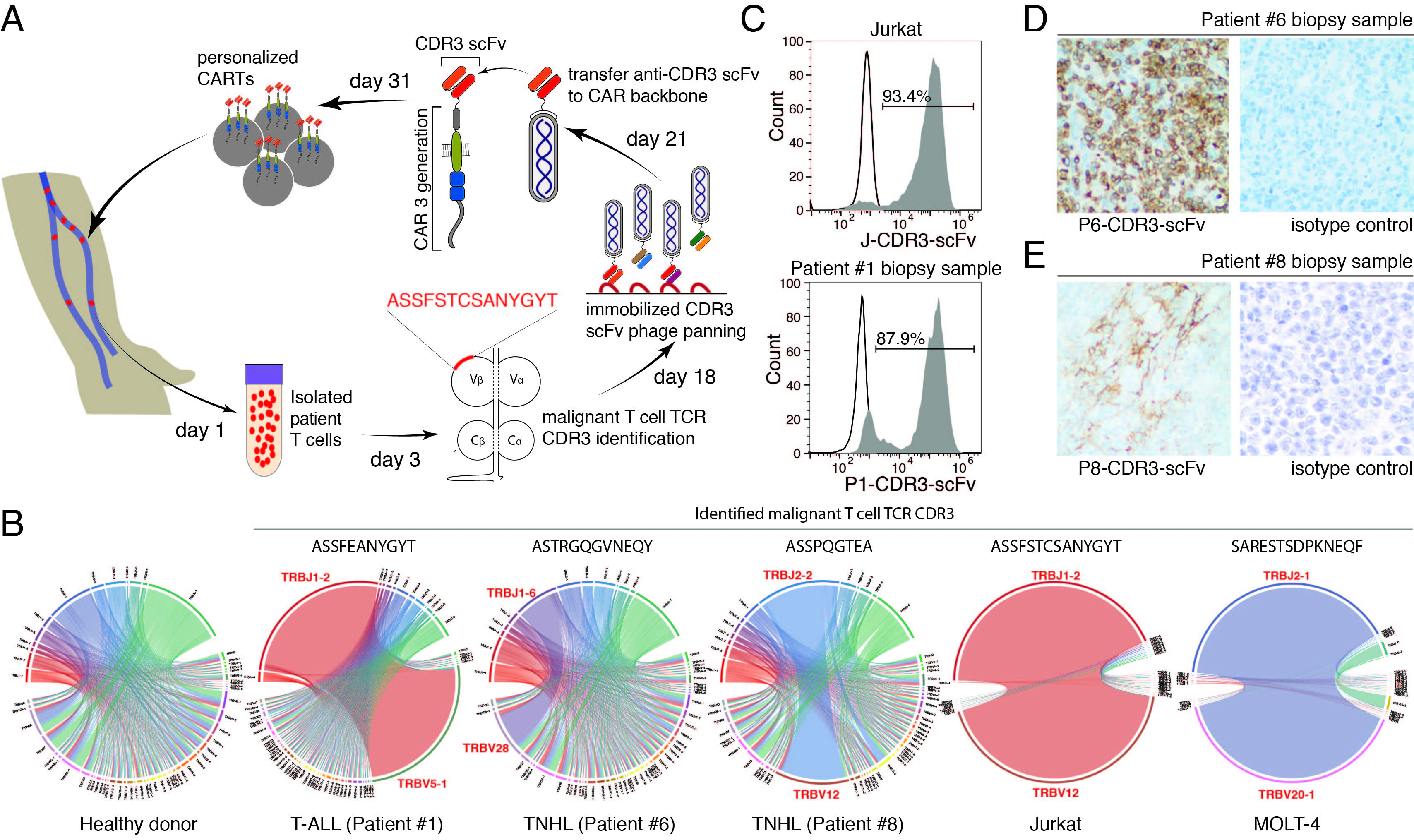
На сегодняшний день не существует CART терапии для элиминации раковых лимфоцитов Т-клеточного происхождения. Основной проблемой создания такой терапии является поиск уникального антигена, отличающего раковые клетки от здоровых Т-клеток, а также CART клеток. Группа исследователей из ИБХ РАН (Степанов А.В. под руководством академика Габибова А.Г.), научно-исследовательского института Скиппса TSRI (Джинши Хуанг под руководством профессора Джиа Ксие), а также Ксамьенского Университета (Джиан Ли под руководством профессора Чи-Менг Тженга) предложили новый подход к направленной элиминации опухолевых клеток с помощью CARТ-лимфоцитов специфичных к свойственной только для раковых Т-клеток последовательности CDR3 Т-клеточного рецептора. Исследование выполнено при поддержке РНФ и опубликовано в журнале Leukemia.
26 апреля 2019 года

