Пресс-центр / новости / Наука /
Модель пирофосфатного фармакофора: как низин связывает липид II
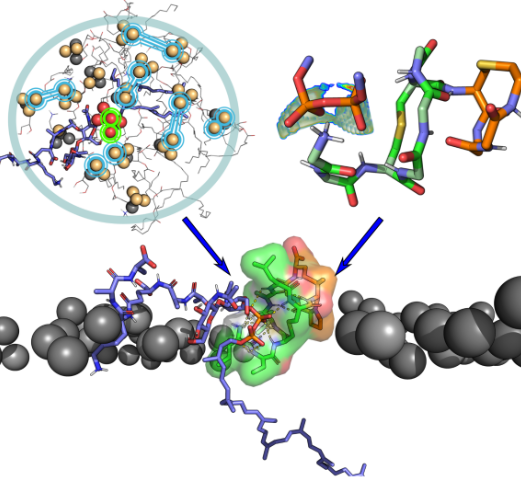
Ученые из Группы анализа структуры мембранных белков in silico и Лаборатории моделирования биомолекулярных систем ИБХ РАН предсказали структуру комплекса лантибиотика низина с предшественником бактериальной клеточной стенки — липидом II — на поверхности мембраны.
С помощью компьютерного моделирования было установлено, что в основе их высокоселективного взаимодействия лежит способность пирофосфатной группы липида II формировать фармакофор на поверхности бислоя, который не может быть образован фосфолипидами: в определенных конформациях атомы кислорода пирофосфата располагаются на поверхности мембраны на уникальном расстоянии. Было показано, что взаимное молекулярное распознавание происходит по механизму индуцированного соответствия и зависит от свойств окружающей среды. Так, среди конформационных ансамблей обоих партнеров было найдено всего одно-два состояния, подходящих для образования комплекса. Эти результаты могут быть полезны для разработки новых антибиотиков на основе низина. Работа опубликована в Scientific Reports.
1 июня 2020 года


