Пресс-центр / новости / Наука /
Hypocrates – новый генетически кодируемый флуоресцентный биосенсор для визуализации (псевдо)гипогалогенных кислот и их производных
Сотрудники Отдела метаболизма и редокс-биологии ИБХ РАН совместно с российскими и иностранными коллегами из Бельгии и Франции разработали новый аналитический инструмент, позволяющий регистрировать (псевдо)гипогалогенный стресс в живых системах в режиме реального времени на субклеточном разрешении. Биосенсор был детально охарактеризован in vitro, в том числе впервые установлена пространственная структура редокс-активного сенсора на основе кругового пермутанта флуоресцентного белка. При помощи полученного инструмента была визуализирована динамика активных форм галогенов в модели воспаления при ампутации хвостового плавника Danio rerio. Работа опубликована в журнале Nature Communications (IF 14.919).

Kostyuk AI, Tossounian M, Panova AS, Thauvin M, Raevskii RI, Ezeriņa D, Wahni K, Van Molle I, Sergeeva AD, Vertommen D, Gorokhovatsky AY, Baranov MS, Vriz S, Messens J, Bilan DS & Belousov VV
В последние годы был накоплен большой массив данных, убедительно показывающий, что многие хронические заболевания человека сопровождаются воспалительной компонентой, для которой характерна инфильтрация активированных нейтрофилов в очаг патологии. Данные клетки обладают специализированными системами ферментов для продукции (псевдо)гипогалогенных кислот – малых неорганических молекул с высокой реакционной активностью, способных ковалентно модифицировать большинство биологических соединений. В норме организм использует эти вещества для борьбы с патогенами, однако, в тех случаях, когда их синтез выходит из-под контроля, они инициируют повреждение собственных тканей и органов. До настоящего момента исследования метаболизма (псевдо)гипогалогенных кислот значительно осложнялись малыми временами жизни изучаемых агентов, что препятствовало их прямой визуализации. Традиционные подходы, основанные на регистрации вторичных продуктов гипогалогенного стресса, не обладают достаточными чувствительностью и селективностью, а также не могут быть применены к живым организмам в режиме реального времени. В
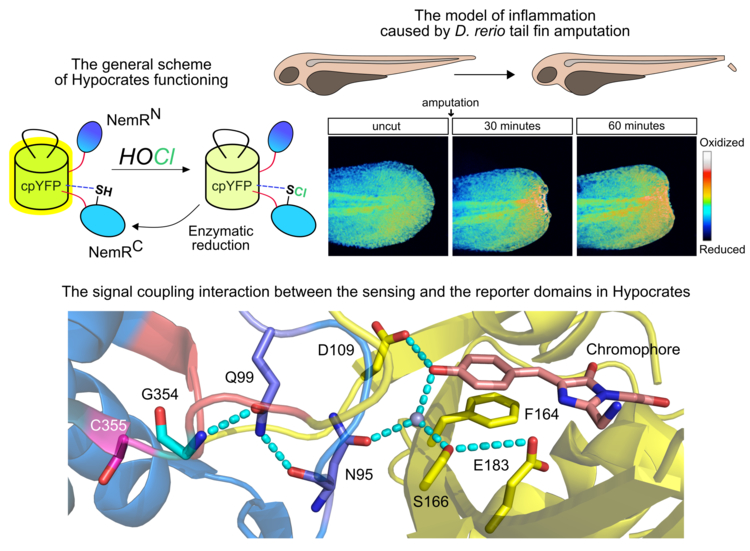
своей работе коллектив авторов разработал генетически кодируемый флуоресцентный сенсор для визуализации (псевдо)гипогалогенных кислот путем интеграции кругового пермутанта желтого флуоресцентного белка (cpYFP) в область транскрипционного репрессора NemR из E. coli. Полученный индикатор селективно взаимодействует с активными формами (псевдо)галогенов (HOCl, HOBr, HOSCN, галамины), что влечет за собой изменение его оптических свойств. Ответ инструмента является обратимым, что позволяет визуализировать не только динамику гипогалогенного стресса, но и работу антиоксидантных систем по его нейтрализации. Биосенсор был назван Hypocrates.
Hypocrates стал первым редокс-индикатором на основе cpFP, для которого расшифрована пространственная структура. Полученная информация проливает свет на механизм сопряжения ключевого остатка цистеина и микроокружения хромофора, что в будущем может быть использовано для направленного улучшения свойств не только обсуждаемого сенсора, но и других химерных белков с аналогичной архитектурой.
При помощи нового инструмента авторы визуализировали динамику гипогалогенного стресса в живых бактериях, фагоцитируемых первичной культурой нейтрофилов человека. Коллектив также зарегистрировал продукцию (псевдо)гипогалогенных кислот в модели ампутации хвостового плавника рыбы D. rerio. Работы, опубликованные ранее, показали, что активность нейтрофилов по генерации HOCl необходима для устранения градиента пероксида водорода, который служит для привлечения лейкоцитов в рану. Таким образом, Hypocrates позволил впервые пронаблюдать данный процесс в режиме реального времени.
12 января 2022 года

