Пресс-центр / новости / Наука /
Синтез рибозидов 2-хлорпурина с амидами хиральных аминокислот в положении С6 и их оценка в качестве агонистов аденозиновых рецепторов А1 типа
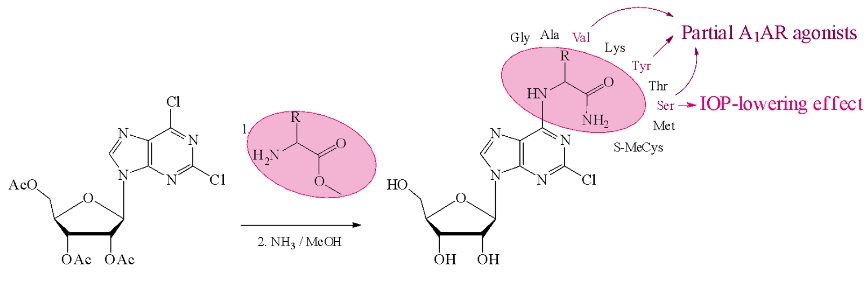
Команда ученых из Отдела биотехнологии и Лаборатория структурной биологии ионных каналов ИБХ РАН синтезировала серию новых аналогов аденозина – производных 2-хлорпурин-рибозида, содержащих в положении С6 амиды аминокислот и провела молекулярный докинг по их сродству к аденозиновым рецепторам типа А1 (A1AR).
Коллегами из ВолгГМУ (Волгоград) было проведено исследовано стимулирующей A1AR активности на модели изолированного предсердия мыши. Показано, что производные с остатками тирозина, валина и серина проявляют свойства частичных агонистов A1AR. Эксперименты на животных в тесте «открытое поле» показали, что эти соединения имеют разные профили психоактивного действия. Производное с сериновым заместителем обладает офтальмологическим гипотензивным эффектом. Синтезированные нуклеозиды могут стать основой для дальнейшего дизайна и синтеза новых агонистов A1AR. Работа опубликована в журнале Bioorganic Chemistry.
14 июня 2022 года


