Press-room / news / Science news /
Chemists discovered a new mechanism of action of a Vipera nikolskii venom neurotoxin
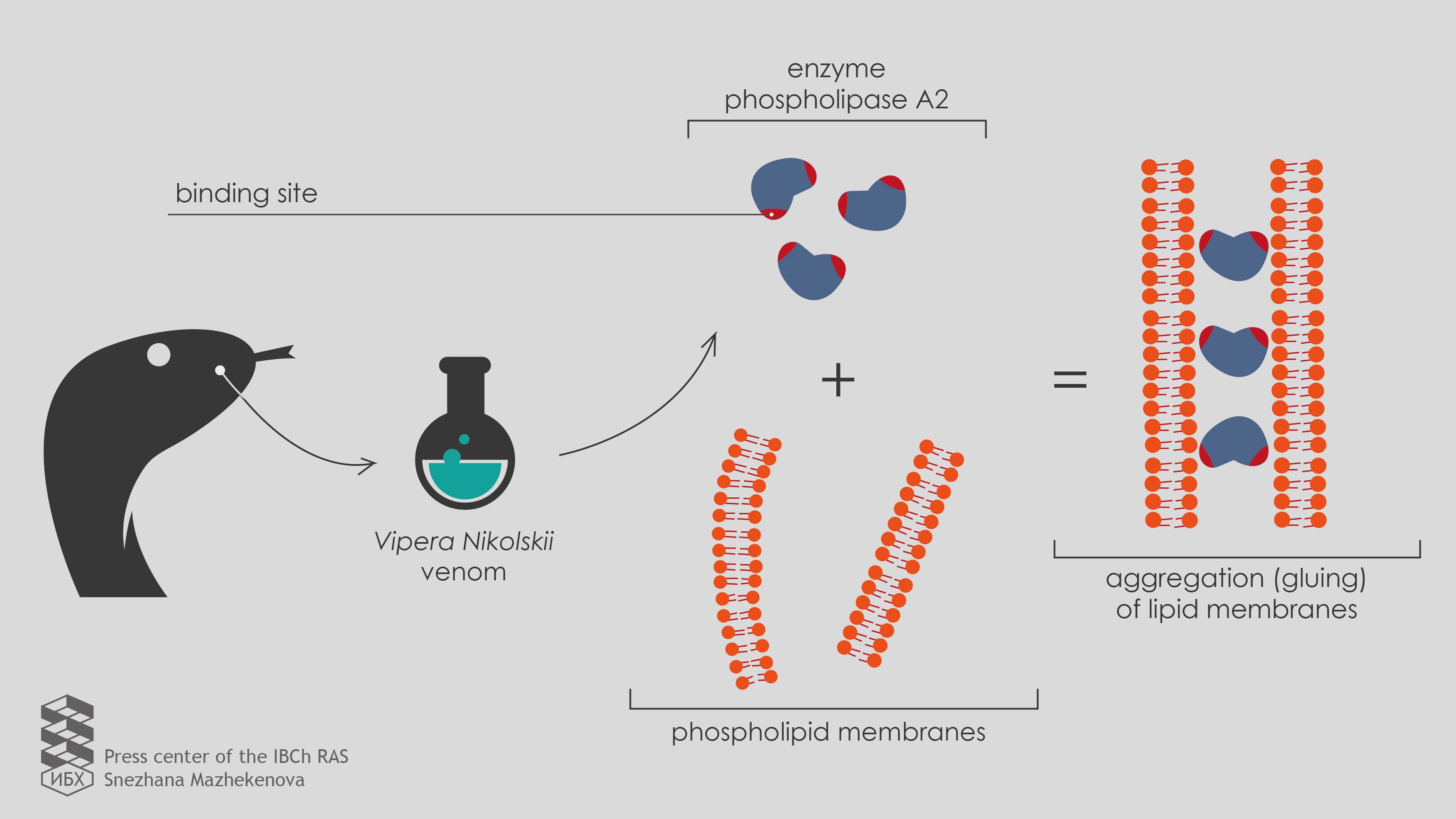
Scientists from the Shemyakin-Ovchinnikov Institute of Bioorganic Chemistry RAS (IBCh RAS) with the participation of colleagues from the Institute of Human Morphology of RAMS and the Pirogov RSMU found that the enzyme phospholipase A2 from the Vipera nikolskii venom is able to adhere to lipid membranes (prototypes of cell membranes) and cause their aggregation, even if the activity of enzyme is specifically blocked. It turned out that this is due to the presence of two binding sites in the enzyme structure. Published in the journal Toxicon the results of the work help to understand how the multicomponent snakes’ venoms work.
Animals are protected from dangers in many ways. Some avoid a death in meeting with enemy with help of camouflage, others – with claws, teeth or hooves. And, for example, some insects, spiders, scorpions and snakes have poisons that can not only paralyze, but also kill the attacker. Many researchers are interested in the poisons’ components, because they can be used to produce antidotes and create new drugs. They contain neurotoxins – poisons, which act on the nervous system and block the conducting of a nerve impulse. These neurotoxins include certain types of enzyme phospholipase A2.
The researchers’ interest to phospholipase A2 is due to the fact that it serves as a marker of inflammation. This means that the level of this enzyme in the blood rises with acute inflammation in the body. Therefore, several scientific groups in the world are trying to create systems for detecting the activity of phospholipase A2.
In 2016, the Laboratory of Lipid Chemistry of IBCh RAS stuff have developed a fundamentally new fluorescence method for detecting the interaction of this enzyme with a double molecular layer of lipids (the so-called lipid bilayer) – the basic structure of the cell membrane. The method is based on energy transfer between two fluorescent dyes (fluorophores attached to the molecules of phospholipids) embedded in the lipid bilayer.
“To test a new development on as many objects as possible, we turned to the Laboratory of Molecular Toxinology IBCh RAS,” says Ivan Boldyrev, a senior researcher at the Laboratory of Lipid Chemistry. “The head of the Laboratory Yuri Utkin have collected a set of phospholipases A2 from poisons of various organisms, including two heterodimeric phospholipases A2 from the Vipera nikolskii venom. Each of these enzymes consists of two heterofunctional subunits, polypeptide chains folded in a specific way. However, the toxic effect mechanism of these heterodimers is not clear.”
The research with the help of the new system gave unexpected results. Under the action of these “double proteins” (heterodimers), fluorescence did not flare up, as occurs with all other phospholipases, but, on the contrary, it decays.
“We realized that we came across something fundamentally new,” Ivan continues. A more in-depth study using electron microscopy showed that heterodimeric phospholipases A2 from the Vipera nikolskii venom cause the aggregation of lipid membranes that means that they “glue” them together. However, this effect emerged only with negatively charged membranes.
“Uncharged membranes which have no electric charge on the surface do not combine under the action of heterodimers,” explains Anna Alekseeva, a junior researcher at the Laboratory of Lipid Chemistry.
“We managed to establish the specificity of heterodimeric phospholipases A2 for negatively charged membranes and determine the conditions of pH of the medium at which the enzyme manifests itself,” adds DariaTretyakova, a PhD-student at the Laboratory of Lipid Chemistry.
The new data help to understand the effect of multicomponent snake venoms, and the developed methodology will be in demand for studying other lipid-protein interactions.
june 8, 2017

