Пресс-центр / новости / Наука /
В ИБХ РАН получило развитие новое направление синтетической химии
Низкомолекулярные соединения играют важнейшую роль во многих биологических процессах и используются в роли лекарственных средств и иных инструментов биологических исследований. Появление новых классов подобных веществ невозможно без разработки новых методов синтеза.
В результате совместной работы, проведенной Лабораторией биоинформационных методов комбинаторной химии и биологии и Группой гетероциклических соединений при технической поддержке Лаборатории биомолекулярной ЯМР-спектроскопии, был разработан новый уникальный метод синтеза спироциклических производных, основанный на использовании нового класса циклопропанов. Результат исследования опубликован в журнале Organic Letters.
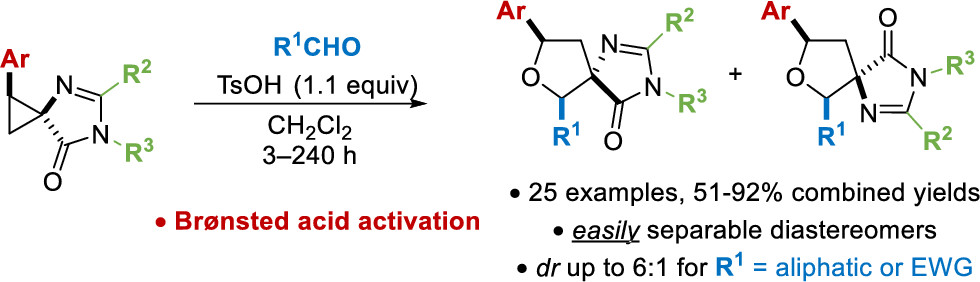
Спироаннелированные циклопропаны, содержащие фрагмент имидазол-5-она, были исследованы в реакции с альдегидами с образованием тетрагидрофуранового цикла. Изученные соединения могут быть рассмотрены как новый вариант донорно-акцепторных циклопропанов - популярных в последнее время билдинг-блоков в органическом синтезе, выступающих в качестве 1,3-цвиттерионных синтонов.
Cпиро[имидазол-5-он-тетрагидрофураны], продукты реакции, являются оригинальными производными альфа-аминокислот. Спироциклическое строение делает их привлекательными объектами в дизайне лекарственных средств.
20 марта 2020 года


