Пресс-центр / новости / Наука /
Многопараметрические вычисления позволяют создавать биологические антидоты против фосфорорганических соединений с высокой реакционной способностью и заданной стереоселективностью
Сотрудниками Лаборатории биокатализа, Лаборатории химии протеолитических ферментов, Лаборатории биоинформационных методов комбинаторной химии и биологии и Лаборатории белков гормональной регуляции, совместно с коллегами из ЕМБЛ-Гамбург, МГУ им. М.В. Ломоносова, Института Шеффилда и Исследовательского института Скриппса разработан универсальный алгоритм, позволяющий создавать биологические антидоты на основе биокатализаторов, направлено увеличивая их реакционную способность, и прогнозировать стереоселективность.
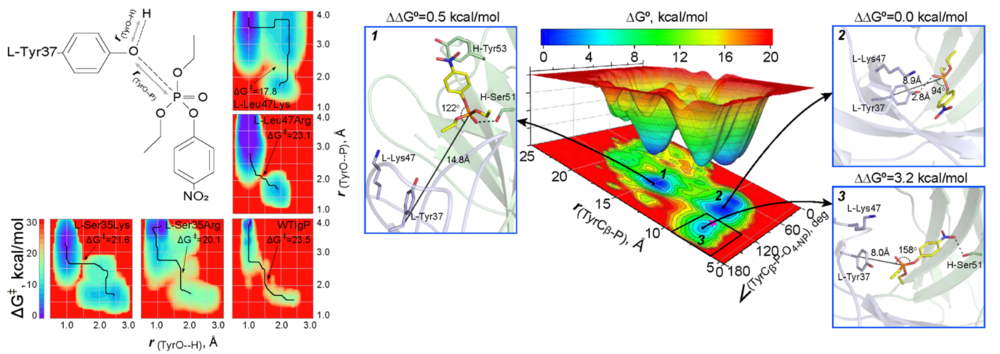
Использование искусственных библиотек генов антител имеет важное преимущество – возможность отбора связывающих и каталитических антител к высокотоксичным соединениям, что невозможно при использовании классических методов иммунизации животных. С другой стороны, использование существующих систем скрининга комбинаторных библиотек антител ограничено методами отбора, что сужает размеры библиотеки, а также не позволяет осуществлять «созревание» антитела, свойственного живым системам. В этой работе существенно усовершенствован разработанный ранее метод QM/MM «созревания» каталитических антител (Smirnov et al. Science Advances 2016). Нами был разработан алгоритм, в котором использовалась комбинация методов DFT-b и Funnel Metadynamics. Это позволило найти вариант антитела, в котором единственная аминокислотная замена L-Leu47Lys приводит к увеличению реакционной способности по отношению к фосфорорганическому пестициду параоксон в 340 раз. Мы определили механизм и детально описали путь реакции образования ковалентной связи между реакционным остатком Tyr37 антитела и параоксоном. Разработанный алгоритм не только позволяет с высокой точностью определять энергетические параметры реакции, но и предсказывать стереоселективность каталитических антител к заданному хиральному субстрату.
Предложенный в статье алгоритм является универсальным и открывает перспективы для направленного дизайна высокоспецифичных, генетически кодируемых акцепторов фосфорорганических соединений, а также для расширения функциональных свойств белков членов суперсемейства иммуноглобулинов, включая клеточные рецепторы.
Работа опубликована в журнале Proceedings of the National Academy of Sciences.
10 сентября 2020 года




