Пресс-центр / новости / Наука /
От цитоскелета к плюрипотентности: новый механизм регуляции стволового статуса эмбриональных клеток
Сотрудники Лаборатории молекулярных основ эмбриогенеза ИБХ РАН в техническом сотрудничестве с коллегами из Отдела метаболизма и редокс-биологии ИБХ РАН, Лаборатории геномики и эпигеномики позвоночных Федерального центра «Фундаментальные основы биотехнологии» РАН, а также группы клеточной подвижности Института белка РАН, открыли не известный ранее механизм регуляции активности генов, определяющих плюрипотентный статус эмбриональных стволовых клеток.
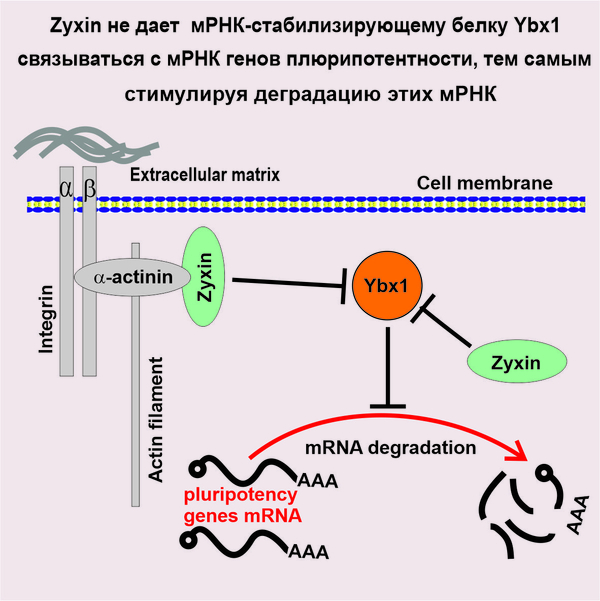
Центральную роль в открытом механизме играет LIM-доменный белок Zyxin. Ранее было известно, что основная функция Zyxin в клетке - регуляция сборки актиновых филаментов на цитоплазматической части клеточных контактов. Помимо этого, Zyxin может перемещаться в ядро, играя роль передатчика сигналов от клеточных контактов к геному. Такое перемещение Zyxin в ядро индуцируется, например, воздействием на клеточные контакты механических напряжений. В связи с этим предполагается, что особенно важную роль Zyxin играет в эмбриональном развитии, когда необходима координация морфогенетических движений клеток, порождающих механические напряжения в тканях, с экспрессией генов. Авторы настоящей работы показали, что подавление активности Zyxin у модельного объекта, эмбрионов шпорцевой лягушки Xenopus laevis, снижает экспрессию генов-регуляторов дифференцировки, но при этом усиливает экспрессию генов, ответственных за статус эмбриональных стволовых клеток. На примере генов семейства pou5f3 (гомологи OCT4 - одного из мастер-генов, определяющих статус эмбриональных стволовых клеток у человека) было установлено, что увеличение концентрации мРНК pou5f3 происходит за счет ее стабилизации в результате образования комплекса с Y-бокс мРНК-связывающим фактором Ybx1. Однако Zyxin, связываясь Ybx1, препятствует образованию комплексов Ybx1 с мРНК pou5f3, что ускоряет ее деградацию. Аналогичное действие Zyxin было выявлено и в отношении мРНК других основных генов плюрипотентности эмбриональных стволовых клеток: Klf4 и Vent1/2 (амфибийные гомологи известного гена плюрипотентности NANOG млекопитающих). В подтверждение универсальности открытого механизма, сходные эффекты Zyxin были продемонстрированы авторами для мРНК генов поддержания эмбрионально-стволового статуса клеток, KLF4, NANOG и OCT4, на таких модельных объектах как эмбрионы рыбы данио и клети эмбриональной почки человека. Все эти данные указывают, что Zyxin играет роль переключателя между стволовым и дифференцированным статусом эмбриональных клеток. Учитывая важную роль Zyxin как регулятора клеточных морфогенетических движений, логично предположить, что с помощью открытого авторами статьи механизма в эмбриогенезе осуществляется координация процессов морфогенеза и клеточной дифференцировки.
Статья опубликована в журнале Cell Reports.
17 ноября 2020 года




