Пресс-центр / новости / Наука /
Тайна свечения огненных червей раскрыта: молекулярные основы биолюминесценции Odontosyllis
Биолюминесценция, «холодный свет», излучаемый живыми организмами, - это широко распространенное природное явление. В его основе лежит катализируемая ферментом реакция окисления низкомолекулярного субстрата, энергия которой высвобождается в виде света. Для более чем 40 известных на данный момент биолюминесцентных систем известны структуры всего 9 субстратов – люциферинов, причем полный путь биосинтеза, для которого были бы известны все ферменты участники, описан лишь для двух из них.
Совместные усилия международной команды ученых (в работе принимали участие Лаборатория химии метаболических путей, Лаборатория лиганд-рецепторных взаимодействий, Лаборатория биомолекулярной ЯМР-спектроскопии и Группа масс-спектрометрии ИБХ РАН) привели к открытию нового люциферина из морской полихеты Odontosyllis undecimdonta. Описание структур трех ключевых низкомолекулярных компонентов биолюминесцентной системы Odontosyllis, представленное в статье, опубликованной в журнале Proceedings of the National Academy of Sciences USA, позволило так же прояснить молекулярные основы биолюминесценции данного организма.
Оказалось, что структуры люциферина Odontosyllis undecimdonta, оксилюциферина, а также продукта неспецифического окисления люциферина кислородом содержат в своей основе крайне необычный трициклический гетероцикл, содержащий три атома серы с различными степенями окисления. Люциферин Odontosyllis, имеющий абсолютно непохожую на другие известные люциферины структуру, позволил исследователям описать новый химический механизм биолюминесценции. Кроме того, оксилюциферин Odontosyllis является единственным из известных для морских люминесцентных организмов первичным эмиттером зеленого света. Представленные открытия позволят в будущем стимулировать развитие новых технологий, инструментов и аналитических методов для различных приложений в биологии и медицине, включая биоимиджинг живых организмов и скрининг лекарственных препаратов.
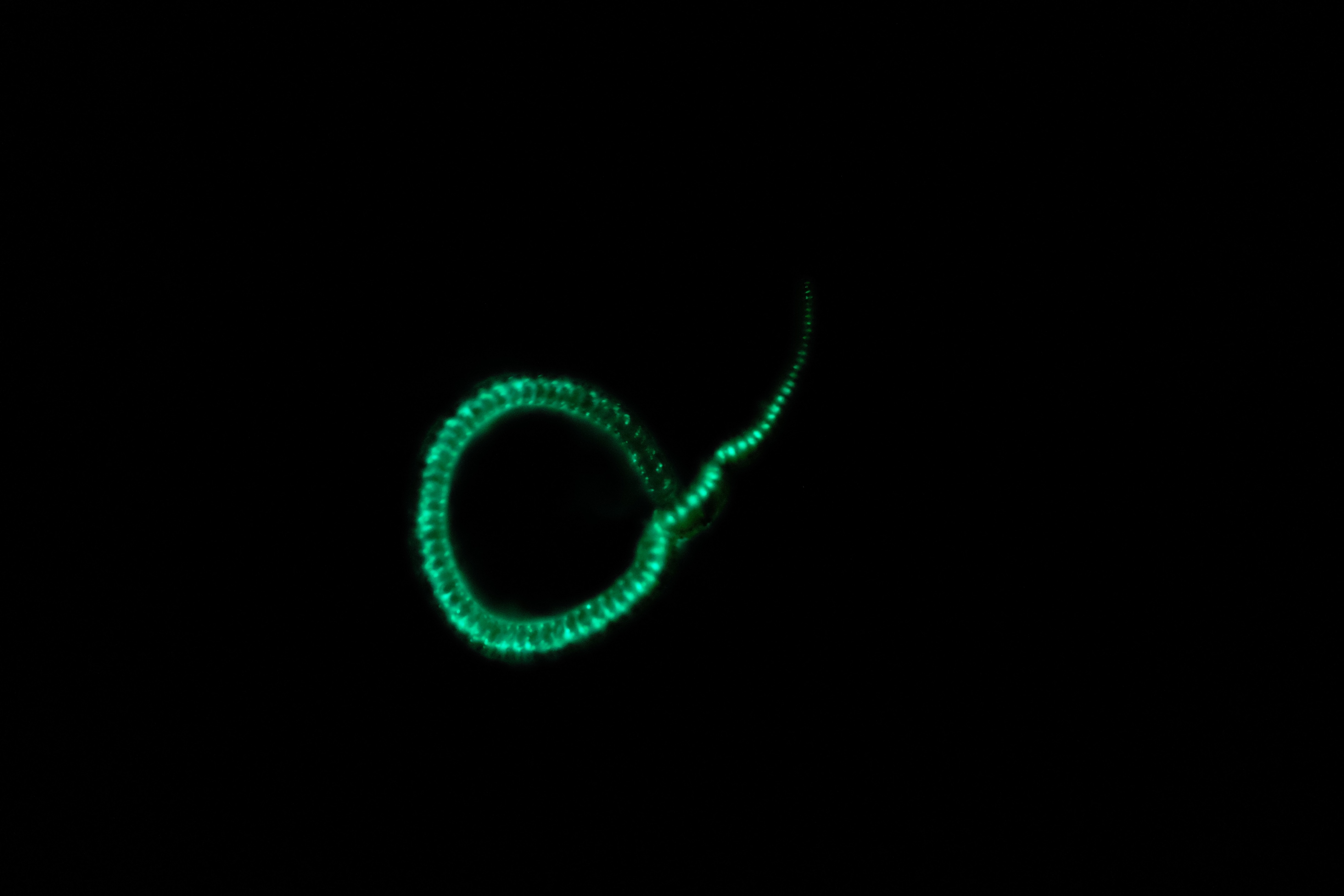

Изображение огненного червя Odontosyllis undecimdonta при дневном освещении и в темноте. Фотографии предоставлены профессором Юичи Оба (Yuichi Oba, Chubu University, Japan).
КОНТАКТЫ ДЛЯ ПРЕССЫ:
Александра Царькова, e-mail: altsarkova@gmail.com; тел.: +7 926 7069715
Илья Ямпольский, e-mail: ivyamp@gmail.com; тел.: +7 916 3227149
Институт Биоорганической Химии РАН, Москва, Россия
29 августа 2019 года

